The Impact of Low-Temperature Storage on Lithium Battery Performance
In extreme cold environments, the internal workings of a lithium-ion battery resemble a slow-moving symphony: ion migration is impeded, interfacial reactions become unbalanced, and performance ultimately declines sharply. As lithium battery engineers, we fully understand the effects of low-temperature storage on battery performance.
In cold conditions, the insertion and extraction of lithium ions within the battery become impeded, reducing the number of available ions. Some ions may even be reduced to metallic lithium, forming dendrites that deposit on the anode surface, causing irreversible capacity loss.
More critically, these lithium dendrites may continue growing until they pierce the separator, triggering internal short circuits that dramatically accelerate battery life degradation.
I. Key Effects of Low-Temperature Environments on Lithium Batteries
Using a temperature-controlled test chamber to simulate low-temperature storage conditions, we conducted systematic research on soft-pack lithium batteries employing the LiFePO₄/graphite system. Testing encompassed critical parameters including battery capacity retention rate, coulombic efficiency, electrochemical impedance spectroscopy (EIS), charge-discharge curves, incremental capacity, and differential voltage curves.
The experiments utilized pouch-type lithium-ion batteries with a rated capacity of 10Ah and an operating voltage range of 2.30-3.65V. These batteries were stored at -40°C for 12 hours, then returned to 25°C for charge-discharge testing. This cycle was repeated 15 times and compared with a control group tested entirely at 25°C.
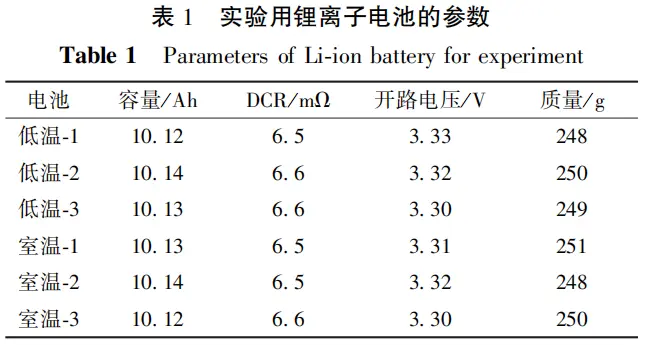
Results revealed that after low-temperature storage, the battery exhibited a 2.04 percentage point decrease in average capacity retention compared to the control group, with an initial coulombic efficiency of only 97.4%—significantly below normal levels.
This indicates that low-temperature storage causes substantial battery aging, with the most pronounced impact occurring during the first exposure to low temperatures.
II. Battery Aging Mechanisms Caused by Low-Temperature Storage
Increased Internal Resistance and Polarization
Electrochemical impedance spectroscopy analysis revealed that the battery’s ohmic resistance increased by 16.18% after low-temperature storage, with a significant rise in charge transfer resistance as well.
The low-temperature environment reduced the activity of the electrolyte and internal battery materials, leading to increased electrolyte viscosity. This disrupted the processes of lithium-ion insertion, extraction, and migration, resulting in decreased ionic conductivity and ultimately manifested as a comprehensive rise in battery internal resistance.
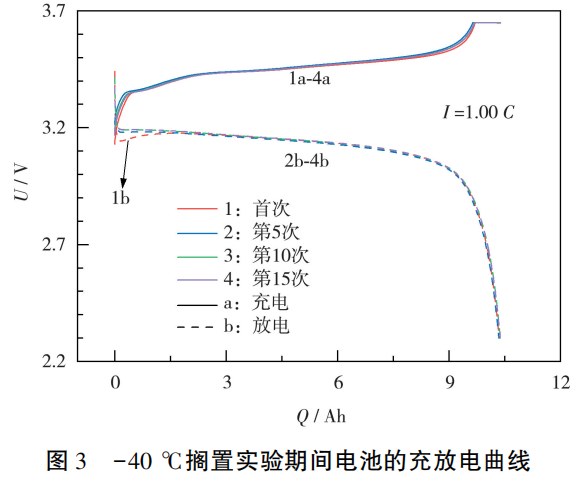
During the initial discharge phase, batteries subjected to low-temperature storage exhibited a pronounced voltage drop, with the discharge voltage temporarily falling to 3.16V before recovering to the normal voltage plateau.
This occurs because, after the first low-temperature storage cycle, some internal materials remain in a “cold shock” state with reduced activity. The active materials fail to fully participate in the reaction, leading to decreased reaction rates and enhanced polarization effects within the battery.
Loss of Active Material and Active Lithium Ions
Analysis of differential voltage curves and incremental capacity curves revealed that characteristic peaks shifted toward lower capacities and voltage plateaus diminished in batteries after low-temperature storage. This indicates irreversible loss of electrode active material.
Simultaneously, characteristic peaks exhibit sharpening, indicating substantial loss of active Li+. The degree of sharpening directly reflects the reduction in active Li+ within the electrode.
This loss of active materials and active Li+ is the fundamental cause of battery capacity degradation.
III. Key Factors Limiting Lithium Battery Performance at Low Temperatures
Ion Transport Kinetic Constraints
At low temperatures, lithium batteries face four major challenges: insufficient ionic conductivity, kinetically constrained charge transfer processes, restricted lithium ion transport across the solid electrolyte interphase (SEI) film, and uncontrolled lithium dendrite growth.
When temperatures drop to -40°C, the diffusion coefficient of Li+ within graphite anodes is approximately 10⁻¹¹ cm²/s. This hinders Li+ insertion into graphite during rapid low-temperature charging, instead causing lithium plating on the anode surface. This triggers dendrite growth, posing significant safety risks.
Interfacial Kinetic Barriers
Recent studies indicate that microscopic processes near the electrode/electrolyte interface, particularly the increased desolvation energy barrier for lithium ions near the solid electrolyte interface at low temperatures and the slow transport of Li⁺ through the SEI, play a decisive role in the low-temperature performance of lithium batteries.
While conventional wisdom attributes limitations to electrolyte conductivity at low temperatures, recent studies suggest that the migration process of lithium ions at the solid-liquid interface may be the primary factor affecting low-temperature performance.
IV. Innovative Strategies for Improving Lithium Battery Low-Temperature Performance
Electrolyte Optimization
Electrolyte design represents the most economical, straightforward, and effective approach to enhancing battery low-temperature performance. Innovative electrolyte formulations must consider:
Low-Viscosity Solvents: Employ a “strong solvent + weak solvent” competitive coordination strategy. For example, DMC disrupts the strong interaction between Li+ and DME, inducing the formation of locally disordered solvation structures and lowering the desolvation energy barrier for Li+.
Highly Soluble Lithium Salts: Select lithium salts with low dissociation energies, such as LiFSI and LiTFSI, to increase free Li⁺ concentration and enhance ionic conductivity.
Interface-Optimizing Additives: Induce the formation of inorganic-rich electrode/electrolyte interface layers to effectively suppress lithium dendrite growth and accelerate interfacial transport kinetics.
Material and Intrinsic Structure Optimization
Through a novel decoupling strategy based on d-orbital engineering, a new class of π-d conjugated coordination polymer materials is constructed. Metal ion centers with specific electronic configurations serve as electrochemically inert structural anchors, stabilizing the entire material framework.
Optimizing the particle size of active materials in both cathodes and anodes is also critical. Research indicates that smaller particle sizes enhance lithium battery performance at low temperatures by shortening the diffusion path for lithium ions between the electrode surface layer and the bulk electrode.
Thermal Management Strategy
From a thermal management perspective, an innovative heating system employs pulsed heat pipes synergistically enhanced with TiO₂ metal oxide nanofluids. The combined preheating and temperature maintenance strategy demonstrates outstanding performance.
This approach achieves a temperature rise rate of up to 3.84°C/min during the preheating phase. In the temperature maintenance phase, the battery reaches dynamic thermal equilibrium with temperature uniformity not exceeding 2.0°C, while maintaining a charge/discharge capacity ratio of at least 96.42%.
Looking ahead, the low-temperature performance of lithium batteries will be significantly enhanced through AI-driven electrolyte design methodologies, advanced in-situ and multidimensional characterization techniques, and the development of novel electrode materials.
By leveraging high-throughput virtual screening of electrolyte components and structure-activity relationship prediction models, we aim to establish quantitative component-performance correlation mechanisms, thereby providing a novel technical pathway for the development of next-generation low-temperature lithium batteries.






