Elemental Composition of Lithium Batteries
From lithium-cobalt-oxygen to silicon-carbon composites, element selection directly determines a lithium battery’s energy density, safety, and cost structure.
As lithium battery engineers, we understand that a battery’s performance is fundamentally determined by its elemental composition. The combination and ratio of different elements create the distinct characteristics of various lithium batteries—from the high energy density demands of consumer electronics to the long-life requirements of energy storage systems, and the balance of safety and performance needed for electric vehicles.
Modern lithium batteries are complex electrochemical systems where element selection not only impacts performance parameters but also directly affects material costs, supply chain security, and environmental sustainability. Understanding the properties and roles of lithium battery element composition is key to mastering the development of lithium battery technology.

I. Cathode Materials: The Key Determinant of Battery Type
Cobalt-Based Cathode Materials
Lithium cobalt oxide (LiCoO₂) was the first commercially available cathode material for lithium batteries. Cobalt provides high operating voltage (3.9V) and energy density. The d-electron orbital properties of cobalt enable structural stability during lithium ion insertion and extraction. However, cobalt’s scarcity and toxicity have driven the development of alternative materials.
Lithium cobalt oxide batteries remain widely used in consumer electronics, achieving mass energy densities up to 240 Wh/kg. However, they exhibit relatively poor thermal stability, and significant cobalt price fluctuations substantially impact costs.
Iron-Based and Manganese-Based Cathodes
Lithium iron phosphate (LiFePO₄) adopts an olivine structure. The strong covalent bonds between iron and phosphorus confer exceptional thermal stability and safety performance to the material. Although its energy density is relatively low (approximately 160 Wh/kg), it offers a cycle life exceeding 6,000 cycles and significant cost advantages.
Lithium manganese oxide (LiMn₂O₄) features a spinel structure that provides three-dimensional lithium-ion diffusion pathways. The natural abundance of manganese keeps its cost low. However, the dissolution effect of manganese can degrade cycle performance, so it is typically blended with other materials.
Ternary Material Systems
NCM (Lithium Nickel Cobalt Manganese Oxide) and NCA (Lithium Nickel Cobalt Aluminum Oxide) balance performance by adjusting the ratio of nickel, cobalt, and manganese (or aluminum). High-nickel formulations (e.g., NCM811) boost energy density to 280Wh/kg, though increased nickel content reduces thermal stability.
Aluminum doping enhances structural stability but complicates manufacturing processes. Current technological trends focus on progressively increasing nickel content while reducing cobalt ratios to balance performance and cost.
II. Anode Materials: The Evolution of Lithium Storage Hosts
Carbon-Based Materials Dominate the Market
Graphite, as the mainstream anode material, offers a theoretical specific capacity of 372 mAh/g due to its layered structure. The success of graphite anodes stems from carbon’s chemical stability, excellent conductivity, and relatively low cost.
The lithium-intercalation mechanism of graphite materials limits volume change to approximately 10% during charge-discharge cycles, ensuring cycling stability. However, graphite capacity has approached its theoretical limit, struggling to meet demands for higher energy density.
The Future Potential of Silicon-Based Anodes
Silicon boasts a theoretical specific capacity of 4200 mAh/g—over ten times that of graphite. Yet, its 300% volume expansion during charge-discharge cycles causes particle pulverization and rapid capacity decay.
Current solutions involve developing silicon-carbon composites, where the carbon matrix buffers volume changes while preserving high capacity. Nanoscale silicon technology and binder optimization further enhance the cycling performance of silicon-based anodes.
Titanium-Based and Other Anodes
Lithium titanate (Li₄Ti₅O₁₂) exhibits spinel structure, delivering exceptional rate capability and ultra-long cycle life (exceeding 20,000 cycles). Titanium’s “zero strain” property enables minimal volume change during cycling. However, its low energy density (175 mAh/g) limits application scope.
III. Electrolyte Systems: Media for Ion Transport
Lithium Salts and Organic Solvents
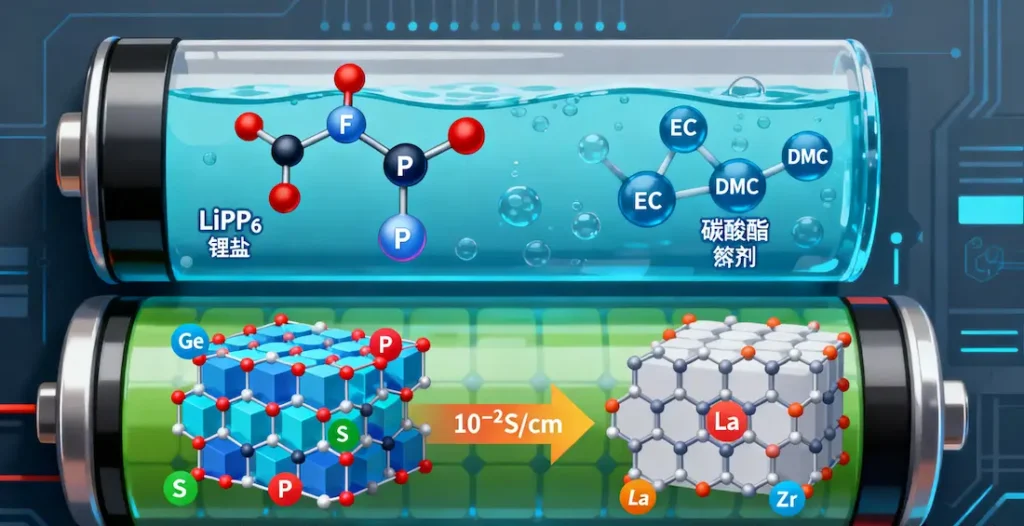
Lithium hexafluorophosphate (LiPF₆) is the current mainstream lithium salt. Its anion, composed of fluorine and phosphorus, exhibits excellent electrochemical stability and moderate ionic conductivity. However, LiPF₆ is sensitive to moisture and decomposes readily at high temperatures.
Organic solvents typically employ carbonate compounds containing carbon, hydrogen, and oxygen. Ethylene carbonate (EC) forms a stable SEI film on graphite anodes, while linear carbonates (e.g., DMC, EMC) reduce viscosity and enhance ion mobility.
Frontiers in Solid-State Electrolytes
Sulfide solid electrolytes (e.g., Li₁₀GeP₂S₁₂) contain elements like germanium, phosphorus, and sulfur, achieving ionic conductivities up to 10⁻² S/cm—approaching liquid electrolyte levels. However, the chemical stability of sulfur remains an unresolved challenge.
Oxide solid-state electrolytes (e.g., LLZO: Li₇La₃Zr₂O₁₂) contain lanthanum, zirconium, and other elements, offering excellent chemical stability but exhibiting high interfacial impedance. Zirconium doping enhances ionic conductivity while suppressing lithium dendrite growth.
IV. Auxiliary Materials and Current Collectors
Separator Function and Materials
Polyolefin separators (polyethylene PE, polypropylene PP) primarily consist of carbon and hydrogen elements. Their microporous structure (pore size below 0.1μm) undergoes a pore closure effect at 130-160°C, providing thermal safety protection. Ceramic-coated separators incorporate elements such as aluminum and oxygen to enhance thermal stability and mechanical strength.
Current Collector Selection Criteria
Aluminum serves as the positive current collector, forming a dense oxide layer on its surface to prevent corrosion. It also offers low density and cost-effectiveness. Copper functions as the negative current collector, exhibiting no alloying reactions at low potentials and delivering excellent electrical conductivity.
Current collectors demand extremely high purity (≥99.9%). Trace impurities catalyze electrolyte decomposition, adversely affecting battery lifespan.
V. Supply, Demand, and Trends of Key Elements
Strategic Element Analysis
Lithium, as a core element, boasts relatively abundant global reserves but exhibits concentrated distribution. Fluctuations in lithium carbonate prices directly impact battery costs. Cobalt’s resource scarcity and supply chain risks drive the development of low-cobalt/cobalt-free technologies.
Nickel demand is growing rapidly with the widespread adoption of high-nickel cathodes, making the supply of high-purity nickel a bottleneck. Copper and aluminum, as traditional industrial metals, offer relatively stable supply but entail high energy consumption.
Emerging Element Potential
Silicon is expanding its application in anodes, supported by the mature photovoltaic-grade silicon supply chain for battery use. Germanium shows promise in solid-state electrolytes, though limited reserves constrain large-scale adoption.
Zirconium stabilizes structures in oxide solid-state electrolytes. While global reserves are ample, purification technology poses significant barriers.
Lithium battery technology is fundamentally the perfect integration of elemental science and electrochemical engineering. From lithium cobalt oxide to lithium iron phosphate, and further to high-nickel ternary and silicon-carbon anodes, each technological advancement has been accompanied by optimization and innovation in element selection.
Current technological development exhibits a trend toward diversification: Lithium iron phosphate solidifies its position in energy storage and the mid-to-low-end electric vehicle market due to its safety and cost advantages; High-nickel ternary materials continue pursuing higher energy density;
Solid-state battery technology is gradually overcoming material interface challenges.
Future element selection for lithium batteries will increasingly prioritize resource sustainability, environmental friendliness, and cost control. Continuous innovation in element combinations will continue pushing the performance boundaries of lithium batteries toward higher energy density, faster charging speeds, longer service life, and enhanced safety standards.







